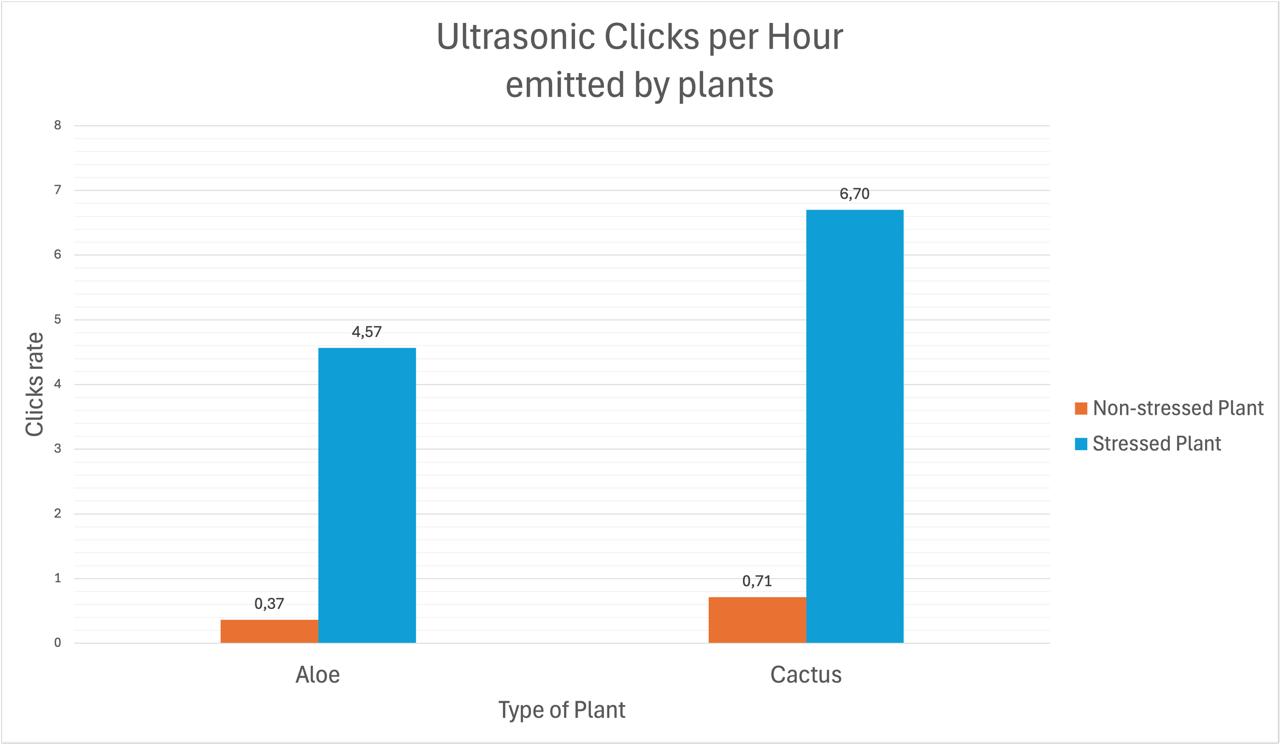
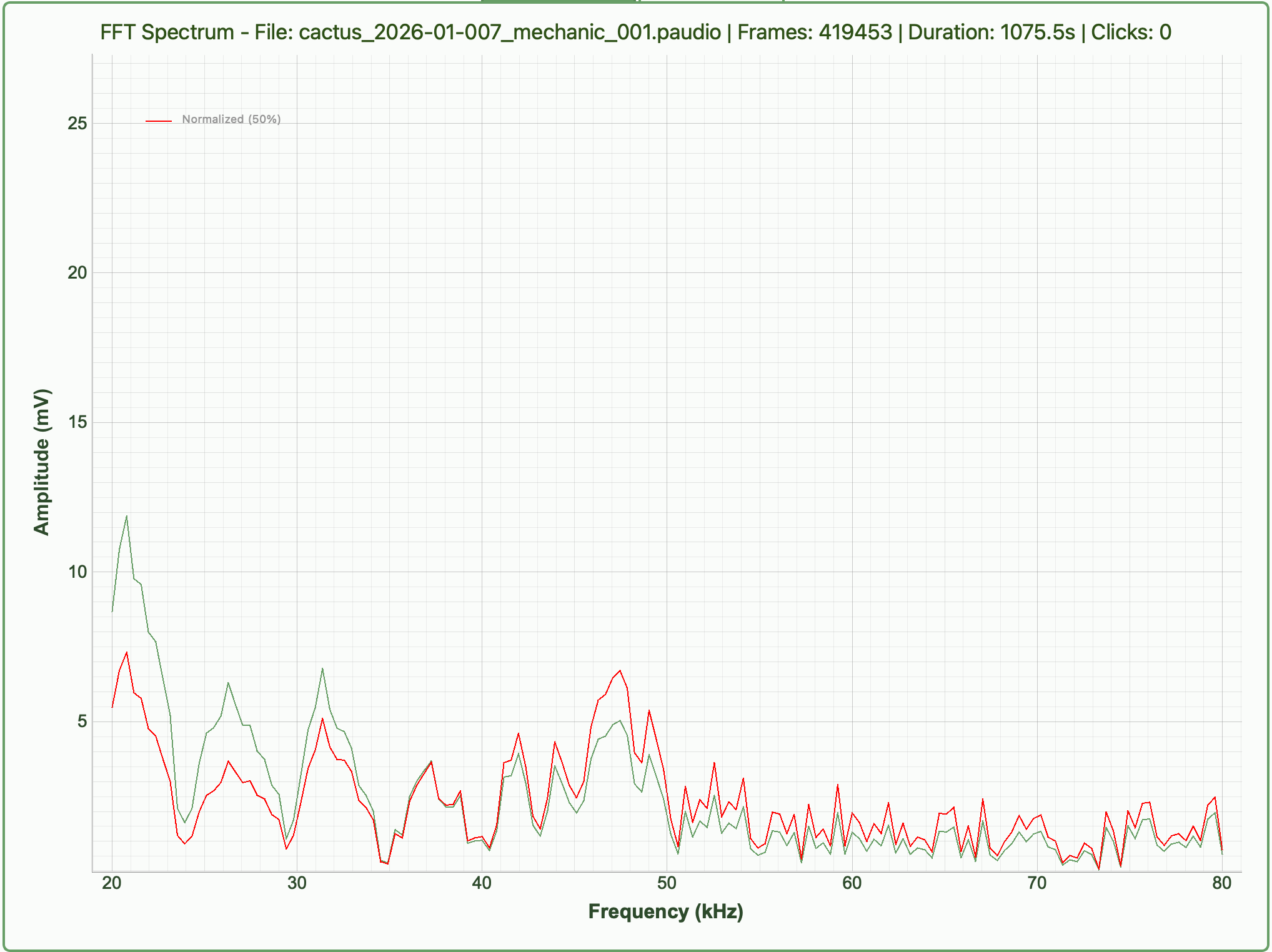
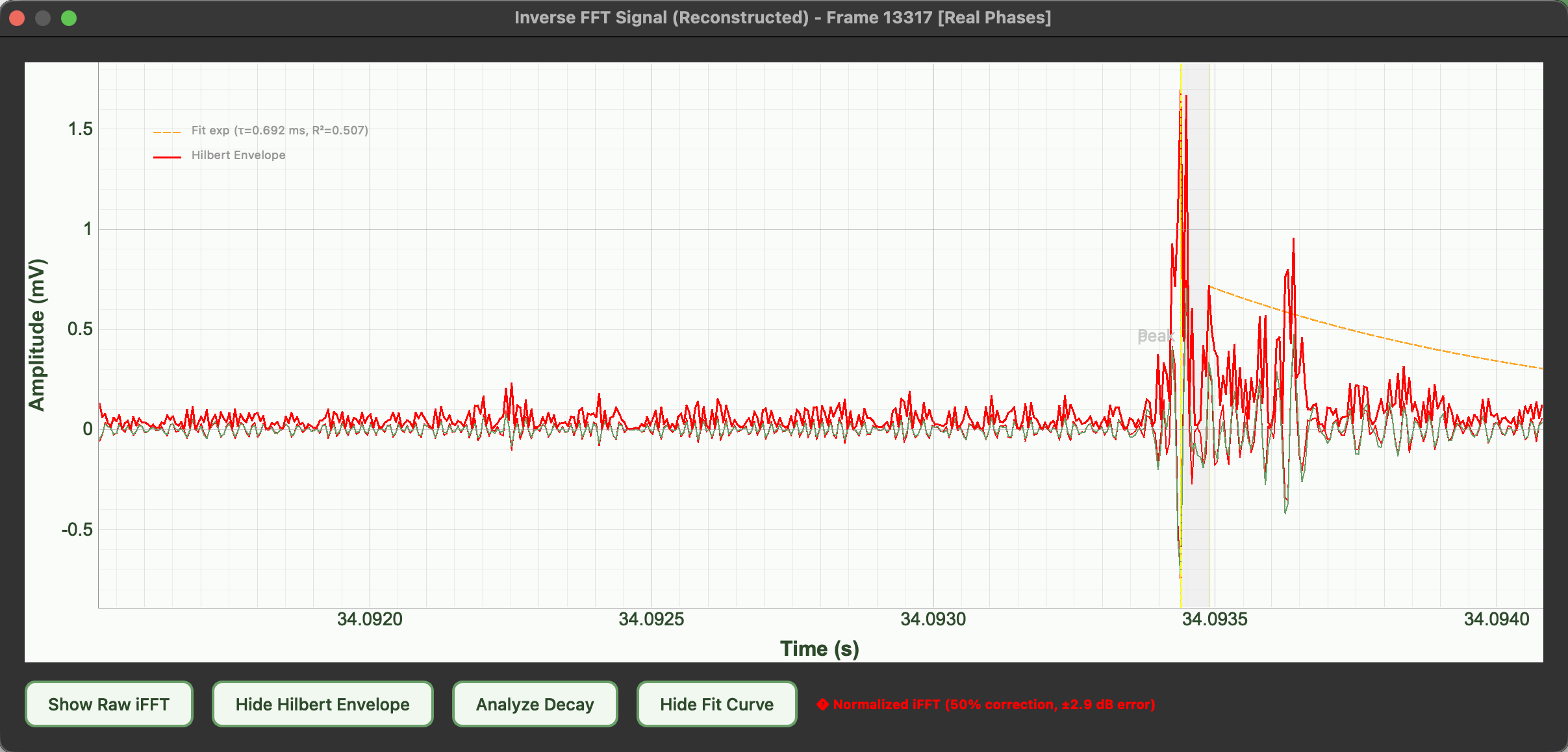
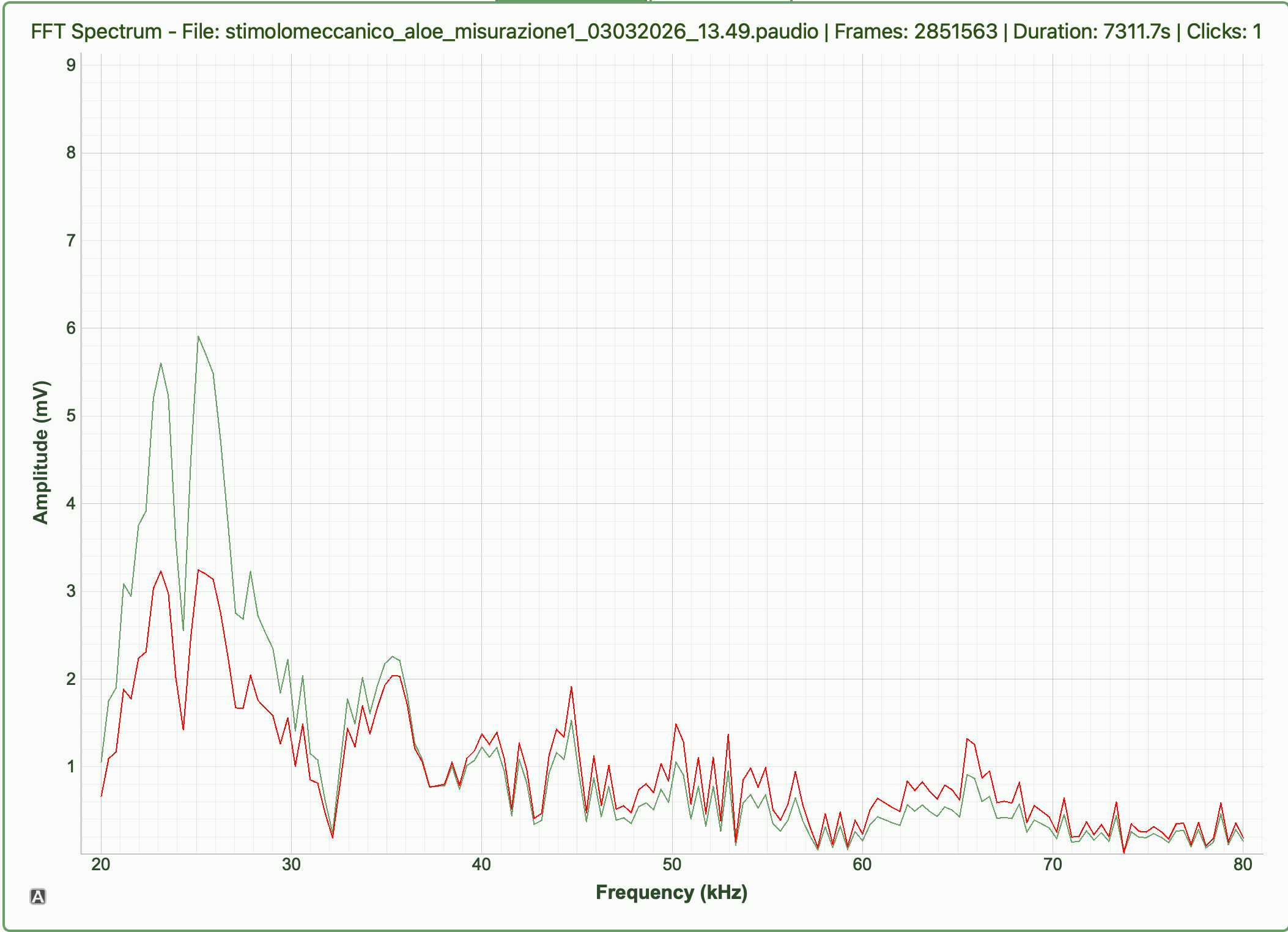
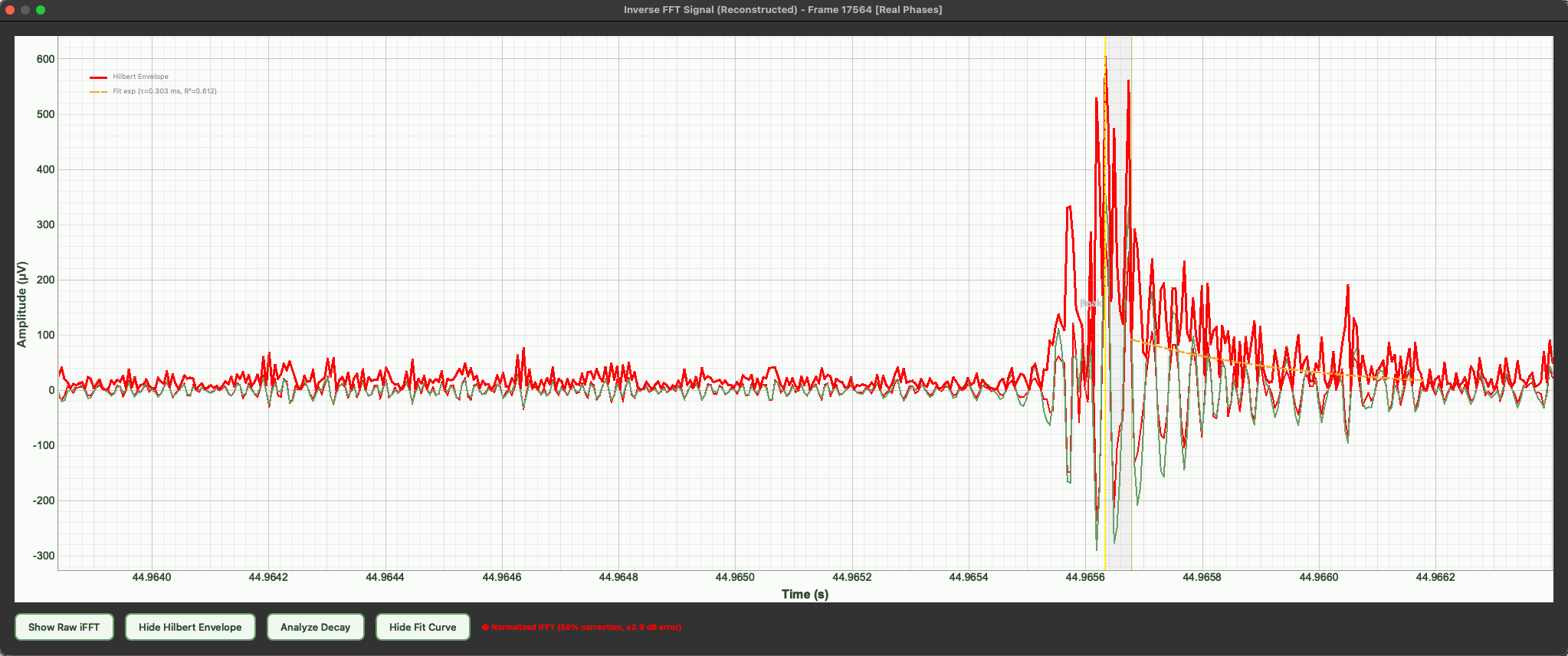
Biological origin of the signals
The emissions are thought to originate from xylem cavitation. During water transport, plants pull water upward through the xylem. Under stress (especially drought), increased tension can form and collapse air bubbles, generating microscopic mechanical vibrations. These vibrations propagate through plant tissue and into the air as ultrasonic clicks. While widely supported, the exact link between cavitation and airborne emissions is still under study.